WARNING - FDA records indicate that this drug has been recalled.
| Product Description: | Cefazolin for Injection USP and Dextrose Injection USP, 2 g in Duplex, 50 mL Container, Catalog Number 3105-11, Rx Only, B Braun Medical Inc., Irvine CA 92614, NDC 0264-3105-11 |
|---|---|
| Status: | Ongoing |
| City: | Irvine |
| State: | CA |
| Country: | US |
| Voluntary/Mandated: | Voluntary: Firm Initiated |
| Initial Firm Notification: | Letter |
| Distribution Pattern: | Nationwide, Puerto Rico and Spain |
| Classification: | Class I |
| Product Quantity: | 42,576 units |
| Reason For Recall: | Presence of Particulate Matter: B. Braun Medical Inc. is recalling several injectable products due to visible particulate matter found in reserve sample units. |
| Recall Initiation Date: | 20131121 |
| Report Date: | 20140507 |
Are you a medical professional?
Trending Topics
Cefazolin Sodium Solution Recall
Get an alert when a recall is issued.
Questions & Answers
Side Effects & Adverse Reactions
There is currently no warning information available for this product. We apologize for any inconvenience.
Legal Issues
There is currently no legal information available for this drug.
FDA Safety Alerts
There are currently no FDA safety alerts available for this drug.
Manufacturer Warnings
There is currently no manufacturer warning information available for this drug.
FDA Labeling Changes
There are currently no FDA labeling changes available for this drug.
Uses
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Cefazolin for Injection USP and Dextrose Injection USP and other antibacterial drugs, Cefazolin for Injection USP and Dextrose Injection USP should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
Cefazolin for Injection USP and Dextrose Injection USP is indicated for the treatment of the following infections when caused by susceptible bacteria.
Respiratory tract infections due to Streptococcus pneumoniae, Staphylococcus aureus and Streptococcus pyogenes.
Injectable benzathine penicillin is considered the drug of choice in treatment and prevention of streptococcal infections, including the prophylaxis of rheumatic fever.
Cefazolin is effective in the eradication of streptococci from the nasopharynx; however, data establishing the efficacy of cefazolin in the subsequent prevention of rheumatic fever are not available.
Urinary tract infections due to Escherichia coli, and Proteus mirabilis.
Skin and skin structure infections due to S. aureus, S. pyogenes, and Streptococcus agalactiae.
Biliary infections due to E. coli, various isolates of streptococci, P. mirabilis, and S. aureus.
Bone and joint infections due to S. aureus.
Genital infections due to E. coli, and P. mirabilis.
Septicemia due to S. pneumoniae, S. aureus, P. mirabilis, and E. coli.
Endocarditis due to S. aureus and S. pyogenes.
The prophylactic administration of cefazolin preoperatively, intraoperatively, and postoperatively may reduce the incidence of certain postoperative infections in patients undergoing surgical procedures which are classified as contaminated or potentially contaminated (e.g., vaginal hysterectomy, and cholecystectomy in high-risk patients such as those older than 70 years, with acute cholecystitis, obstructive jaundice, or common duct bile stones).
The perioperative use of cefazolin may also be effective in surgical patients in whom infection at the operative site would present a serious risk (e.g., during open-heart surgery and prosthetic arthroplasty).
If there are signs of infection, specimens for cultures should be obtained for the identification of the causative organism so that appropriate therapy may be instituted.
History
There is currently no drug history available for this drug.
Other Information
Cefazolin for Injection USP and Dextrose Injection USP is a sterile, nonpyrogenic, single use, packaged combination of Cefazolin Sodium USP (lyophilized) and sterile iso-osmotic diluent in the DUPLEX® sterile container. The DUPLEX® Container is a flexible dual chamber container.
After reconstitution the approximate osmolality for Cefazolin for Injection USP and Dextrose Injection USP is 290 mOsmol/kg.
The drug chamber is filled with sterile lyophilized Cefazolin Sodium USP, a semi-synthetic cephalosporin and has the following IUPAC nomenclature: Sodium (6R,7R)-3-[[(5-methyl-1,3,4-thiadiazol-2-yl)thio]methyl]-8-oxo-7-[2-(1H-tetrazol-1-yl)acetamido]-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylate.
Cefazolin Sodium USP has the following structural formula:
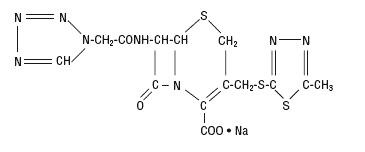
The sodium content is 48 mg/g of cefazolin sodium.
The diluent chamber contains Dextrose Injection USP, an iso-osmotic diluent using Hydrous Dextrose USP in Water for Injection USP. Dextrose Injection USP is sterile, nonpyrogenic, and contains no bacteriostatic or antimicrobial agents.
Hydrous Dextrose USP has the following structural (molecular) formula:
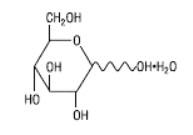
The molecular weight of Hydrous Dextrose USP is 198.17
Cefazolin Sodium USP is supplied as a lyophilized form equivalent to either 1 g or 2 g of cefazolin. Dextrose hydrous USP has been added to the diluent to adjust osmolality (approximately 2 g [4.0% w/v] and 1.5 g [3.0% w/v] for the 1 g and 2 g dosages, respectively).
After removing the peelable foil strip, activating the seals, and thoroughly mixing, the reconstituted drug product is intended for single intravenous use.
The DUPLEX® Container is not manufactured with Latex, PVC or DEHP.
The DUPLEX® dual chamber container is made from a specially formulated material. The product (diluent and drug) contact layer is a mixture of thermoplastic rubber and a polypropylene ethylene copolymer that contains no plasticizers. The safety of the container system is supported by USP biological evaluation procedures.
Sources
Cefazolin Sodium Solution Manufacturers
-
B. Braun Medical Inc.
![Cefazolin Sodium Solution [B. Braun Medical Inc.]](/wp-content/themes/bootstrap/assets/img/loading2.gif)
Cefazolin Sodium Solution | Heritage Pharmaceuticals Inc.
![Cefazolin Sodium Solution [B. Braun Medical Inc.] Cefazolin Sodium Solution [B. Braun Medical Inc.]](/wp-content/themes/bootstrap/assets/img/loading2.gif)
Infusion-related events are related to both the concentration and the rate of administration of vancomycin. Concentrations of no more than 5 mg/mL and rates of no more than 10 mg/min are recommended in adults (see also age-specific recommendations). In selected patients in need of fluid restriction, a concentration up to 10 mg/mL may be used; use of such higher concentrations may increase the risk of infusion related events. An infusion rate of 10 mg/min or less is associated with fewer infusion-related events (see ADVERSE REACTIONS). Infusion-related events may occur, however, at any rate or concentration.
Patients With Normal Renal Function
Adults
The usual daily intravenous dose is 2 g divided either as 500 mg every 6 hours or 1 g every 12 hours.
Each dose should be administered at no more than 10 mg/min, or over a period of at least 60 minutes, whichever is longer. Other patient factors, such as age or obesity, may call for modification of the usual intravenous daily dose.
Pediatric Patients
The usual intravenous dosage of vancomycin is 10 mg/kg per dose given every 6 hours. Each dose should be administered over a period of at least 60 minutes.Close monitoring of serum concentrations of vancomycin may be warranted in these patients.
Neonates
In pediatric patients up to the age of 1 month, the total daily intravenous dosage may be lower. In neonates, an initial dose of 15 mg/kg is suggested, followed by 10 mg/kg every 12 hours for neonates in the 1st week of life and every 8 hours thereafter up to the age of 1 month. Each dose should be administered over 60 minutes. In premature infants, vancomycin clearance decreases as postconceptional age decreases. Therefore, longer dosing intervals may be necessary in premature infants. Close monitoring of serum concentrations of vancomycin is recommended in these patients.
Patients With Impaired Renal Function and Elderly Patients
Dosage adjustment must be made in patients with impaired renal function. In premature infants and the elderly, greater dosage reductions than expected may be necessary because of decreased renal function. Measurement of vancomycin serum concentrations can be helpful in optimizing therapy, especially in seriously ill patients with changing renal function. Vancomycin serum concentrations can be determined by use of microbiologic assay, radioimmunoassay, fluorescence polarization immunoassay, fluorescence immunoassay or high-pressure liquid chromatography.
If creatinine clearance can be measured or estimated accurately, the dosage for most patients with renal impairment can be calculated using the following table. The dosage of vancomycin hydrochloride for injection per day in mg is about 15 times the glomerular filtration rate in mL/min (see following table).
DOSAGE TABLE FOR VANCOMYCIN IN PATIENTS WITH IMPAIRED RENAL FUNCTION
(Adapted from Moellering et al.4)
Creatinine Clearance
Vancomycin Dose
mL/min
mg/24 h
100
1,545
90
1,390
80
1,235
70
1,080
60
925
50
770
40
620
30
465
20
310
10
155
The initial dose should be no less than 15 mg/kg, even in patients with mild to moderate renal insufficiency.
The table is not valid for functionally anephric patients. For such patients, an initial dose of 15 mg/kg of body weight should be given to achieve prompt therapeutic serum concentrations.
The dose required to maintain stable concentrations is 1.9 mg/kg/24 hr. In patients with marked renal impairment, it may be more convenient to give maintenance doses of 250 to 1,000 mg once every several days rather than administering the drug on a daily basis. In anuria, a dose of 1,000 mg every 7 to 10 days has been recommended.
When only serum creatinine is known, the following formula (based on sex, weight and age of the patient) may be used to calculate creatinine clearance. Calculated creatinine clearances (mL/min) are only estimates. The creatinine clearance should be measured promptly.
Men: [Weight (kg) x (140 — age in years)]
72 x serum creatinine concentration (mg/dL)
Women: 0.85 x above value
The serum creatinine must represent a steady state of renal function. Otherwise the estimated value for creatinine clearance is not valid. Such a calculated clearance is an overestimate of actual clearance in patients with conditions: (1) characterized by decreasing renal function, such as shock, severe heart failure or oliguria; (2) in which a normal relationship between muscle mass and total body weight is not present, such as in obese patients or those with liver disease, edema or ascites; and (3) accompanied by debilitation, malnutrition or inactivity.
The safety and efficacy of vancomycin administration by the intrathecal (intralumbar or intraventricular) routes have not been established. Intermittent infusion is the recommended method of administration.
Compatibility with Other Drugs and IV Fluids
The following diluents are physically and chemically compatible (with 4 g/L vancomycin hydrochloride):
5% Dextrose Injection, USP
5% Dextrose Injection and 0.9% Sodium Chloride Injection, USP
Lactated Ringer's Injection, USP
5% Dextrose and Lactated Ringer's Injection
Normosol®-M and 5% Dextrose
0.9% Sodium Chloride Injection, USP
Isolyte® E
Good professional practice suggests that compounded admixtures should be administered as soon after preparation as is feasible.
Vancomycin solution has a low pH and may cause physical instability of other compounds.
Mixtures of solutions of vancomycin and beta-lactam antibiotics have been shown to be physically incompatible. The likelihood of precipitation increases with higher concentrations of vancomycin. It is recommended to adequately flush the intravenous lines between the administration of these antibiotics. It is also recommended to dilute solutions of vancomycin to 5 mg/mL or less.
Although intravitreal injection is not an approved route of administration for vancomycin, precipitation has been reported after intravitreal injection of vancomycin and ceftazidime for endophthalmitis using different syringes and needles. The precipitates dissolved gradually, with complete clearing of the vitreous cavity over two months and with improvement of visual acuity.
PREPARATION AND STABILITY
Directions for Proper Use of a Pharmacy Bulk Package
DIRECTIONS FOR PROPER USE OF PHARMACY BULK PACKAGE
Not for direct infusion. The pharmacy bulk package is for use in the Pharmacy Admixture Service only in a suitable work area such as a laminar flow hood. Using aseptic technique, the closure may be penetrated only one time after reconstitution using a suitable sterile transfer device or dispensing set, which allows measured dispensing of the contents. Use of a syringe and needle is not recommended as it may cause leakage. After entry use entire contents of the Pharmacy Bulk Package bottle promptly. The entire contents of the Pharmacy Bulk Package bottle should be dispensed within 4 hours after initial entry. A maximum time of 4 hours from the initial entry may be allowed to complete fluid aliquoting/transferring operations. Discard the container no later than 4 hours after initial closure puncture. This time limit should begin with the introduction of solvent or diluent into the Pharmacy Bulk Package bottle.
Preparation and Stability
10 g Pharmacy Bulk Package bottle
At the time of use, reconstitute by adding 95 mL of Sterile Water for Injection, USP to the 10 g bottle of dry, sterile vancomycin powder. The resultant solution will contain vancomycin equivalent to 500 mg/5 mL (1 g/10 mL). AFTER RECONSTITUTION, FURTHER DILUTION IS REQUIRED.
Reconstituted solutions of vancomycin (500 mg/5 mL) must be further diluted in at least 100 mL of a suitable infusion solution. For doses of 1 gram (10 mL), at least 200 mL of solution must be used. The desired dose diluted in this manner should be administered by intermittent IV infusion over a period of at least 60 minutes.
Parenteral drug products should be visually inspected for particulate matter and discoloration prior to administration, whenever solution and container permit.
For Oral Administration
Oral vancomycin is used in treating antibiotic -associated pseudomembranous colitis caused by C. difficile and for staphylococcal enterocolitis. Vancomycin is not effective by the oral route for other types of infections. The usual adult total daily dosage is 500 mg to 2 g given in 3 or 4 divided doses for 7 to 10 days. The total daily dose in children is 40 mg/kg of body weight in 3 or 4 divided doses for 7 to 10 days. The total daily dosage should not exceed 2 g. The appropriate dose may be diluted in 1 oz of water and given to the patient to drink. Common flavoring syrups may be added to the solution to improve the taste for oral administration. The diluted solution may be administered via a nasogastric tube.
Login To Your Free Account


