Cefprozil Recall
Get an alert when a recall is issued.
Questions & Answers
Side Effects & Adverse Reactions
BEFORE THERAPY WITH CEFPROZIL IS INSTITUTED, CAREFUL INQUIRY SHOULD BE MADE TO DETERMINE WHETHER THE PATIENT HAS HAD PREVIOUS HYPERSENSITIVITY REACTIONS TO CEFPROZIL, CEPHALOSPORINS, PENICILLINS, OR OTHER DRUGS. IF THIS PRODUCT IS TO BE GIVEN TO PENICILLIN-SENSITIVE PATIENTS, CAUTION SHOULD BE EXERCISED BECAUSE CROSS-SENSITIVITY AMONG ß-LACTAM ANTIBIOTICS HAS BEEN CLEARLY DOCUMENTED AND MAY OCCUR IN UP TO 10% OF PATIENTS WITH A HISTORY OF PENICILLIN ALLERGY. IF AN ALLERGIC REACTION TO CEFPROZIL OCCURS, DISCONTINUE THE DRUG. SERIOUS ACUTE HYPERSENSITIVITY REACTIONS MAY REQUIRE TREATMENT WITH EPINEPHRINE AND OTHER EMERGENCY MEASURES, INCLUDING OXYGEN, INTRAVENOUS FLUIDS, INTRAVENOUS ANTIHISTAMINES, CORTICOSTEROIDS, PRESSOR AMINES, AND AIRWAY MANAGEMENT, AS CLINICALLY INDICATED.
Clostridium difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including cefprozil, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin-producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibiotic use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
Legal Issues
There is currently no legal information available for this drug.
FDA Safety Alerts
There are currently no FDA safety alerts available for this drug.
Manufacturer Warnings
There is currently no manufacturer warning information available for this drug.
FDA Labeling Changes
There are currently no FDA labeling changes available for this drug.
Uses
Cefprozil for oral suspension is indicated for the treatment of patients with mild to moderate infections caused by susceptible strains of the designated microorganisms in the conditions listed below:
Upper Respiratory Tract:
Pharyngitis/Tonsillitis:
Caused by Streptococcus pyogenes.
NOTE: The usual drug of choice in the treatment and prevention of streptococcal infections, including the prophylaxis of rheumatic fever, is penicillin given by the intramuscular route. Cefprozil is generally effective in the eradication of Streptococcus pyogenes from the nasopharynx; however, substantial data establishing the efficacy of cefprozil in the subsequent prevention of rheumatic fever are not available at present.
Otitis Media:
Caused by Streptococcus pneumoniae, Haemophilus influenzae (including ß-lactamase-producing strains), and Moraxella (Branhamella) catarrhalis (including ß-lactamase-producing strains). (See CLINICAL STUDIES.)
NOTE: In the treatment of otitis media due to ß-lactamase producing organisms, cefprozil had bacteriologic eradication rates somewhat lower than those observed with a product containing a specific ß-lactamase inhibitor. In considering the use of cefprozil, lower overall eradication rates should be balanced against the susceptibility patterns of the common microbes in a given geographic area and the increased potential for toxicity with products containing ß-lactamase inhibitors.
Acute Sinusitis:
Caused by Streptococcus pneumoniae, Haemophilus influenzae (including ß-lactamase producing strains), and Moraxella (Branhamella) catarrhalis (including ß-lactamase-producing strains).
Lower Respiratory Tract:
Secondary Bacterial Infection of Acute Bronchitis and Acute Bacterial Exacerbation of Chronic Bronchitis:
Caused by Streptococcus pneumoniae, Haemophilus influenzae (including ß-lactamase-producing strains), and Moraxella (Branhamella) catarrhalis (including ß-lactamase-producing strains).
Skin And Skin Structure:
Uncomplicated Skin and Skin-Structure Infections:
Caused by Staphylococcus aureus (including penicillinase producing strains) and Streptococcus pyogenes. Abscesses usually require surgical drainage.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of cefprozil for oral suspension and other antibacterial drugs, cefprozil for oral suspension should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
History
There is currently no drug history available for this drug.
Other Information
Cefprozil is a semi-synthetic broad-spectrum cephalosporin antibiotic.
Cefprozil is a cis and trans isomeric mixture (≥90% cis). The chemical name for the monohydrate is (6R,7R)-7-[(R)-2-amino-2-(p-hydroxyphenyl)acetamido]-8-oxo-3-propenyl-5-thia-1-azabicyclo [4.2.0]oct-2-ene-2- carboxylic acid monohydrate, and the structural formula is:
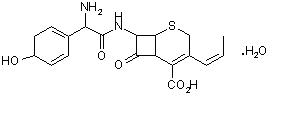
Cefprozil is a white to yellowish powder with a molecular formula for the monohydrate of C18H19N3O5S.H2O and a molecular weight of 407.45.
Cefprozil for oral suspension is intended for oral administration.
Cefprozil for oral suspension contains cefprozil equivalent to 125 mg or 250 mg of anhydrous cefprozil per 5 mL constituted suspension. In addition, the oral suspension contains the following inactive ingredients: aspartame, bubble gum flavor, anhydrous citric acid, colloidal silicon dioxide, FD&C Red No. 40 Aluminum Lake, glycine, microcrystalline cellulose and sodium carboxymethylcellulose, sodium benzoate, sodium chloride and sucrose.
Sources




![Cefprozil (Cefprozil) Powder, For Suspension [Lupin Pharmaceuticals Inc]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=3ec55c0a-1456-412e-972f-352b3135c8f2&name=cefprozil-figure-02.jpg)
![Cefprozil Tablet [Redpharm Drug Inc.]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=0f96e4fe-8daf-492b-8fd8-90ff25243077&name=6729605901.jpg)
![Cefprozil (Cefprozil) Tablet [H. J. Harkins Company, Inc.]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=17a1975e-ce3b-4057-ae5b-8d191319caa7&name=label.jpg)
![Cefprozil Tablet [Physicians Total Care, Inc.]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=75d57e35-ffeb-4d88-8dba-5458e284fd82&name=5756.jpg)
![Cefprozil Tablet, Film Coated [Teva Pharmaceuticals Usa Inc]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=56a507f3-0cd1-4f02-9dfd-de297ba4bc1f&name=5ad7f9da-722f-4280-9278-1f41edce0dfb-01.jpg)
![Cefprozil Powder, For Suspension [Teva Pharmaceuticals Usa Inc]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=6737ed97-9559-44c7-889b-6bc0e20eb36b&name=a23a103f-43ff-4e2e-8e0c-7b0a01794d10-02.jpg)
![Cefprozil Tablet, Film Coated Cefprozil Tablet, Film Coated [Wockhardt Usa Llc.]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=f85f23ef-a73f-47f7-8283-8dab1afdc792&name=cefproziltablets-figure-02.jpg)
![Cefprozil Tablet, Film Coated Cefprozil Tablet, Film Coated [Wockhardt Limited]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=225011b9-0f3a-4afc-ac80-bd1f3739bb3b&name=cefproziltablets-figure-02.jpg)
![Cefprozil Powder, For Suspension [Northstar Rx Llc]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=69d3782b-a5ca-468e-aa7f-05c1eefc9770&name=CefprozilOS125mg50ml.jpg)
![Cefprozil Tablet [Lupin Pharmaceuticals, Inc.]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=14527f4a-ce99-449d-a5a5-9edd7fb28dee&name=cefprozil-tab-figure-02.jpg)
![Cefprozil Powder, For Suspension [Northstar Rx Llc]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=8e3943b5-8c30-4c81-bcfa-aeaaa1d75fb4&name=cefprozil-fig1.jpg)
![Cefprozil Tablet, Film Coated [Northstar Rx Llc]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=6133611e-19c1-4df2-8c23-8573aaf63020&name=cefprozil-fig1.jpg)
![Cefprozil Tablet, Film Coated [Apotex Corp]](http://recallguide.cwdevelopsp.com/wp-content/themes/recallguide/assets/img/drug-image-placeholder.jpg)
![Cefprozil (Cefprozil) Powder, For Suspension [A-s Medication Solutions Llc]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=4d17e35a-0ca1-4a6b-8b69-ea8f0d62938f&name=5785-0.jpg)
![Cefprozil Powder, For Suspension [Aurobindo Pharma Limited]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=97675251-70b8-43bc-93ea-f9ef6bb8cb68&name=cefprozil-fig1.jpg)
![Cefprozil Tablet, Film Coated [Aurobindo Pharma Limited]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=b9111a81-07c4-4458-b994-a4ad504986d8&name=cefprozil-fig1.jpg)
![Cefprozil Tablet, Film Coated [Citron Pharma Llc]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=af24a86f-8072-49a6-94ec-2f17fe81812b&name=cefprozil-fig1.jpg)
![Cefprozil Powder, For Suspension [Citron Pharma Llc]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=d7fac9cc-30e1-4762-a01b-04f1bd3b2a56&name=cefprozil-fig1.jpg)
![Cefprozil Tablet, Film Coated Cefprozil Powder, For Suspension [Dava Pharmaceuticals Inc]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=d2dbfc48-063b-41c0-864c-2c9426ba89c1&name=d2dbfc48-063b-41c0-864c-2c9426ba89c1-02.jpg)
![Cefprozil Tablet, Film Coated Cefprozil Powder, For Suspension [Sandoz Inc]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=a547cc0a-6af0-49c7-a5ea-c41684cc36ae&name=993312ad-d759-41ff-9020-55adb30781c5-02.jpg)
![Cefprozil Powder, For Suspension [Orchidpharma Inc]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=9825b787-72de-4555-9907-3738bd761e57&name=CefprozilOS125mg50ml.jpg)
![Cefprozil Tablet, Film Coated [Medsource Pharmaceuticals]](http://dailymed.nlm.nih.gov/dailymed/image.cfm?setid=1fbe6b78-80fe-223b-e054-00144ff88e88&name=716-20.jpg)