Enablex Recall
Get an alert when a recall is issued.
Questions & Answers
Side Effects & Adverse Reactions
There is currently no warning information available for this product. We apologize for any inconvenience.
Legal Issues
There is currently no legal information available for this drug.
FDA Safety Alerts
There are currently no FDA safety alerts available for this drug.
Manufacturer Warnings
There is currently no manufacturer warning information available for this drug.
FDA Labeling Changes
There are currently no FDA labeling changes available for this drug.
Uses
ENABLEX® (darifenacin) extended-release tablets are indicated for the treatment of overactive bladder with symptoms of urge urinary incontinence, urgency and frequency.
History
There is currently no drug history available for this drug.
Other Information
ENABLEX® (darifenacin) is an extended-release tablet which contains 7.5 mg or 15 mg darifenacin as its hydrobromide salt. The active moiety, darifenacin, is a potent muscarinic receptor antagonist.
Chemically, darifenacin hydrobromide is (S)-2-{1-[2-(2,3-dihydrobenzofuran-5-yl)ethyl]-3-pyrrolidinyl}-2,2-diphenylacetamide hydrobromide. The empirical formula of darifenacin hydrobromide is C28H30N2O2.HBr.
The structural formula is
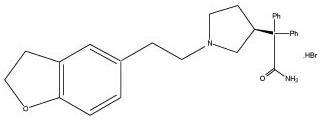
Darifenacin hydrobromide is a white to almost white, crystalline powder, with a molecular weight of 507.5.
ENABLEX is a once a day extended release tablet and contains the following inactive ingredients: dibasic calcium phosphate, hypromellose (hydroxypropyl methylcellulose), magnesium stearate, polyethylene glycol, talc, titanium dioxide. The 15-mg tablet also contains iron oxide red and iron oxide yellow.
Sources