FDA records indicate that there are no current recalls for this drug.
Are you a medical professional?
Trending Topics
Rexall Vanishing Scent Pain Relieving Recall
Get an alert when a recall is issued.
Questions & Answers
Side Effects & Adverse Reactions
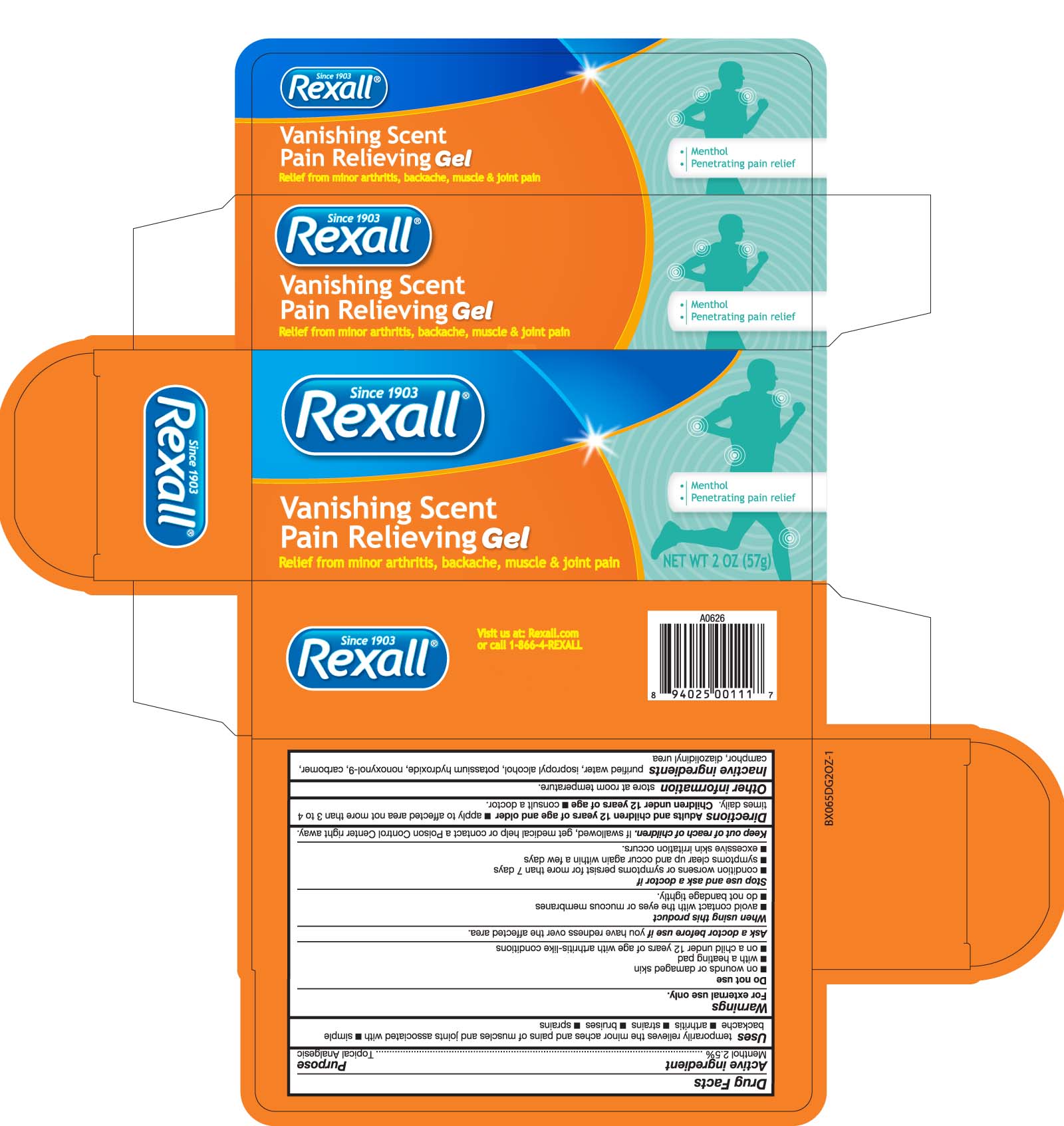
Warnings
For external use only
Do not use
- on wounds or damaged skin
- with a heating pad
- on a child under 12 years of age with arthritis-like conditions
Ask a doctor before use if you have redness over the affected area.
When using this product
- avoid contact with the eyes or mucous membranes
- do not bandage tightly
Stop use and ask a doctor if
- condition worsens or symptoms persist for more than 7 days
- symptoms clear up and occur again within a few days
- excessive skin irritation occurs.
Keep out of reach of children. If swallowed, get medical help or contact a Poison
Control Center right away.
Legal Issues
There is currently no legal information available for this drug.
FDA Safety Alerts
There are currently no FDA safety alerts available for this drug.
Manufacturer Warnings
There is currently no manufacturer warning information available for this drug.
FDA Labeling Changes
There are currently no FDA labeling changes available for this drug.
Uses
Uses temporarily relieves the minor aches and pains of muscles and joints associated with
- simple backache - atrhritis - strains - bruises - sprains
History
There is currently no drug history available for this drug.
Other Information
There are no additional details available for this product.
Sources
Rexall Vanishing Scent Pain Relieving Manufacturers
-
Dolgencorp, Llc
Login To Your Free Account