FDA records indicate that there are no current recalls for this drug.
Are you a medical professional?
Trending Topics
Vivelle-dot Recall
Get an alert when a recall is issued.
Questions & Answers
Side Effects & Adverse Reactions
There is currently no warning information available for this product. We apologize for any inconvenience.
Legal Issues
There is currently no legal information available for this drug.
FDA Safety Alerts
There are currently no FDA safety alerts available for this drug.
Manufacturer Warnings
There is currently no manufacturer warning information available for this drug.
FDA Labeling Changes
There are currently no FDA labeling changes available for this drug.
Uses
Vivelle-Dot is indicated for:
Limitation of Use
When prescribing solely for the treatment of moderate to severe symptoms of vulvar and vaginal atrophy, topical vaginal products should be considered.
Limitation of Use
When prescribing solely for the prevention of postmenopausal osteoporosis, therapy should only be considered for women at significant risk of osteoporosis and non-estrogen medications should be carefully considered.
History
There is currently no drug history available for this drug.
Other Information
Vivelle-Dot (estradiol transdermal system) contains estradiol in a multipolymeric adhesive. The system is designed to release estradiol continuously upon application to intact skin.
Five dosage strengths of Vivelle-Dot are available to provide nominal in vivo delivery rates of 0.025, 0.0375, 0.05, 0.075, or 0.1 mg of estradiol per day via the skin. Each corresponding system has an active surface area of 2.5, 3.75, 5.0, 7.5, or 10.0 cm2 and contains 0.39, 0.585, 0.78, 1.17, or 1.56 mg of estradiol USP, respectively. The composition of the systems per unit area is identical.
Estradiol USP is a white, crystalline powder, chemically described as estra-1,3,5 (10)- triene-3,17β-diol.
The structural formula is:

The molecular formula of estradiol is C18H2402. The molecular weight is 272.39.
Vivelle-Dot is comprised of 3 layers. Proceeding from the visible surface toward the surface attached to the skin, these layers are (1) a translucent polyolefin film (2) an adhesive formulation containing estradiol, acrylic adhesive, silicone adhesive, oleyl alcohol, NF, povidone, USP and dipropylene glycol, and (3) a polyester release liner which is attached to the adhesive surface and must be removed before the system can be used.
 |
----- (1) Backing ----- (2) Adhesive Containing Estradiol ----- (3) Protective Liner |
The active component of the system is estradiol. The remaining components of the system are pharmacologically inactive.
Sources
Vivelle-dot Manufacturers
-
Novartis Pharmaceuticals Corporation
![Vivelle-dot (Estradiol) Patch, Extended Release [Novartis Pharmaceuticals Corporation]](/wp-content/themes/bootstrap/assets/img/loading2.gif)
Vivelle-dot | Novartis Pharmaceuticals Corporation
![Vivelle-dot (Estradiol) Patch, Extended Release [Novartis Pharmaceuticals Corporation] Vivelle-dot (Estradiol) Patch, Extended Release [Novartis Pharmaceuticals Corporation]](/wp-content/themes/bootstrap/assets/img/loading2.gif)
Generally, when estrogen is prescribed for a postmenopausal woman with a uterus, a progestin should be considered to reduce the risk of endometrial cancer. A woman without a uterus does not need a progestin. In some cases, however, hysterectomized women with a history of endometriosis may need a progestin [see Warnings and Precautions (5.2, 5.14)].
Use of estrogen-alone or in combination with a progestin, should be with the lowest effective dose and the shortest duration consistent with treatment goals and risks for the individual woman. Postmenopausal women should be reevaluated periodically as clinically appropriate to determine whether treatment is still necessary.
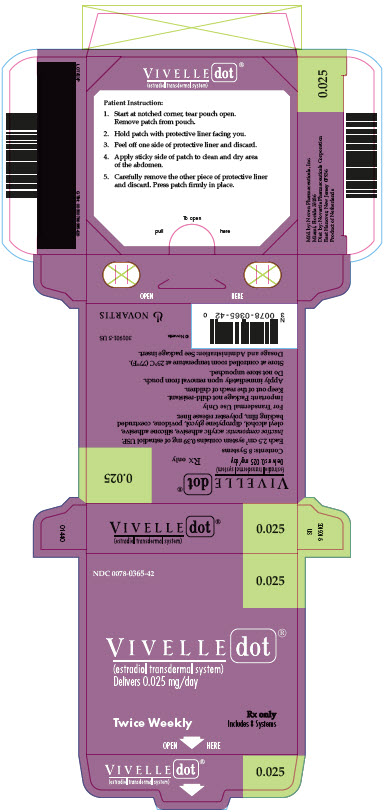
The adhesive side of Vivelle-Dot should be placed on a clean, dry area of the trunk of the body (including the abdomen or buttocks). Vivelle-Dot should not be applied to the breasts.
Vivelle-Dot should be replaced twice weekly. The sites of application must be rotated, with an interval of at least 1 week allowed between applications to a particular site. The area selected should not be oily, damaged, or irritated. The waistline should be avoided, since tight clothing may rub the system off. The system should be applied immediately after opening the pouch and removing the protective liner. The system should be pressed firmly in place with the palm of the hand for about 10 seconds, making sure there is good contact, especially around the edges. In the event that a system should fall off, the same system may be reapplied. If the same system cannot be reapplied, a new system should be applied to another location. In either case, the original treatment schedule should be continued. If a woman has forgotten to apply a patch, she should apply a new patch as soon as possible. The new patch should be applied on the original treatment schedule. The interruption of treatment in women taking Vivelle-Dot might increase the likelihood of breakthrough bleeding, spotting and recurrence of symptoms.
2.1 Moderate to Severe Vasomotor SymptomsStart therapy with Vivelle-Dot 0.0375 mg per day applied to the skin twice weekly. Dosage adjustment should be guided by the clinical response. In women not currently taking oral estrogens or in women switching from another estradiol transdermal therapy, treatment with Vivelle-Dot may be initiated at once. In women who are currently taking oral estrogens, treatment with Vivelle-Dot should be initiated 1 week after withdrawal of oral hormone therapy, or sooner if menopausal symptoms reappear in less than 1 week. Attempts to taper or discontinue the medication should be made at 3 to 6 month intervals.
Vivelle-Dot may be given continuously in patients who do not have an intact uterus. In those patients with an intact uterus, Vivelle-Dot may be given on a cyclic schedule (for example, 3 weeks on drug followed by 1 week off drug).
2.2 Moderate to Severe Symptoms of Vulvar and Vaginal AtrophyStart therapy with Vivelle-Dot 0.0375 mg per day applied to the skin twice weekly. Dosage adjustment should be guided by the clinical response. Attempts to taper or discontinue the medication should be made at 3 to 6 month intervals. In women not currently taking oral estrogens or in women switching from another estradiol transdermal therapy, treatment with Vivelle-Dot may be initiated at once. In women who are currently taking oral estrogens, treatment with Vivelle-Dot should be initiated 1 week after withdrawal of oral hormone therapy, or sooner if menopausal symptoms reappear in less than 1 week.
Vivelle-Dot may be given continuously in patients who do not have an intact uterus. In those patients with an intact uterus, Vivelle-Dot may be given on a cyclic schedule (for example, 3 weeks on drug followed by 1 week off drug).
2.3 Hypoestrogenism Due to Hypogonadism, Castration, or Primary Ovarian Failure 2.4 Prevention of Postmenopausal OsteoporosisStart therapy with Vivelle-Dot 0.025 mg per day applied to the skin twice weekly.
In women not currently taking oral estrogens or in women switching from another estradiol transdermal therapy, treatment with Vivelle-Dot may be initiated at once. In women who are currently taking oral estrogens, treatment with Vivelle-Dot should be initiated 1 week after withdrawal of oral hormone therapy, or sooner if menopausal symptoms reappear in less than 1 week.
Vivelle-Dot may be given continuously in patients who do not have an intact uterus. In those patients with an intact uterus, Vivelle-Dot may be given on a cyclic schedule (for example, 3 weeks on drug followed by 1 week off drug).
Login To Your Free Account